Docusate
 Docusate sodium | |
| Clinical data | |
|---|---|
| Trade names | Colace, Ex-Lax Stool Softener, others |
| Other names | Dioctyl sulfosuccinate |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a601113 |
| License data | |
| Pregnancy category |
|
| Routes of administration | By mouth, rectal |
| Drug class | Stool softener |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Onset of action | 12 hrs to 5 days[1] |
| Duration of action | 3 days[1] |
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| DrugBank |
|
| ChemSpider | |
| UNII |
|
| KEGG |
|
| ChEBI |
|
| ChEMBL |
|
| E number | E480 (thickeners, ...) |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.008.553 |
| Chemical and physical data | |
| Formula | C20H37O7S |
| Molar mass | 421.57 g·mol−1 |
| 3D model (JSmol) |
|
| Density | 1.1 g/cm3 |
| Melting point | 153 to 157 °C (307 to 315 °F) 173-179 °C |
| Solubility in water | 1 in 70 parts mg/mL (20 °C) |
| |
| |
Docusate is the common chemical and pharmaceutical name of the anion bis(2-ethylhexyl) sulfosuccinate, also commonly called dioctyl sulfosuccinate (DOSS).[2][3][4]
Salts of this anion, especially docusate sodium, are widely used in medicine as laxatives and as stool softeners, by mouth or rectally.[1] Some studies claim that docusate is not more effective than a placebo for improving constipation.[5][6][7][8] Other docusate salts with medical use include those of calcium and potassium.[9][1][2] Docusate salts are also used as food additives, emulsifiers, dispersants, and wetting agents, among other uses.[10]
It is on the World Health Organization's List of Essential Medicines.[11] In 2022, it was the 148th most commonly prescribed medication in the United States, with more than 3 million prescriptions.[12][13] In 2022, the combination with senna was the 261st most commonly prescribed medication in the United States, with more than 1 million prescriptions.[12][14]
History
[edit]Sodium docusate was patented in 1937 by Coleman R. Caryl and Alphons O. Jaeger for American Cyanamid,[3] which commercialized it for many years as a detergent under the brand name Aerosol OT.
Its use for the treatment of constipation was first proposed in 1955 by James L. Wilson and David G. Dickinson,[4] and quickly popularized under the name Doxinate.[15]
Medical use
[edit]Constipation
[edit]The main medical use of docusate sodium is to treat constipation, acting as a laxative and stool softener. In painful anorectal conditions such as hemorrhoid and anal fissures, it can help avoid pain caused by straining during bowel movements.
When administered by mouth, a bowel movement often occurs in 1 to 3 days,[1] while rectal use may be effective within 20 minutes.[16]
Sodium docusate is recommended as a stool softener for children.[1]
However, its effectiveness for constipation is poorly supported by evidence.[5][6] Multiple studies have found docusate to be no more effective than a placebo for improving constipation.[5][6][7][8] Others have found it to be less useful for the treatment of chronic constipation than psyllium.[8][17][18]
The medication may be given to people who are receiving opioid medication, although prolonged use may cause irritation of the gastrointestinal tract.[8][18]
Other medical uses
[edit]Docusate sodium, when used with ear syringing, may help with earwax removal, particularly in the case of impaction.[19]
Sodium docusate is also used as a lubricant in the production of tablets and as an emulsifier in topical preparations and other suspensions.[20]
Precautions and contraindications
[edit]Docusate sodium is approved and recommended as safe during pregnancy and breastfeeding.[21][22]
Docusate is not recommended in people with appendicitis, acute abdomen, or ileus.[18]
When taken by mouth it should be ingested with plenty of water.
Side effects
[edit]Side effects are uncommon and typically mild,[1] and may include stomach pain, abdominal cramps or diarrhea,[1] Efficacy decreases with long-term use, and may cause poor bowel function.[9]
Serious allergic reactions may occur with the drug. The most severe side effect of docusate, although very rare, is rectal bleeding.[23]
Interactions
[edit]Docusate might increase resorption of other drugs, for example, dantron (1,8-dihydroxyanthraquinone).[18]
Mechanism of action
[edit]Docusate is an anionic surfactant, which works by reducing the surface tension of the stool, allowing more intestinal water and fat to combine with the stool.[24][9][25] This decreases the strain and discomfort associated with constipation.[24]
It does not stay in the gastrointestinal tract, but is absorbed into the bloodstream and excreted via the gallbladder[18] after undergoing extensive metabolism.
The effect of docusate may not necessarily be all due to its surfactant properties. Perfusion studies suggest that docusate inhibits fluid absorption or stimulates secretion in the portion of the small intestine known as the jejunum.[medical citation needed]
Pharmaceutical brand names
[edit]In the U.S., docusate sodium for pharmaceutical use is available under multiple brand names: Aqualax, Calube, Colace, Colace Micro-Enema, Correctol Softgel Extra Gentle, DC-240, Dialose, Diocto, Dioctocal, Dioctosoftez, Dioctyn, Dionex, Doc-Q-Lace, Docu Soft, Docucal, Doculax, Docusoft S, DOK, DOS, Doss-Relief, DSS, Dulcolax - Stool Softener (not to be confused with another drug marketed under the Dulcolax brand, bisacodyl, which is a stimulant laxative), Ex-Lax Stool Softener, Fleet Sof-Lax, Genasoft, Kasof, Laxa-basic, Modane Soft, Octycine-100, Pedia-Lax, Preferred Plus Pharmacy Stool Softener, Regulax SS, Sulfalax Calcium, Sur-Q-Lax, Surfak Stool Softener, and Therevac-SB. Generic preparations are also available.
In the UK, dioctyl sodium sulfosuccinate is sold under the brand name Docusol (Typharm Ltd) and DulcoEase (Boehringer Ingelheim).
In Australia, dioctyl sodium sulfosuccinate is sold as Coloxyl and Coloxyl with senna.
In India, preparations include Laxatin by Alembic, Doslax by Raptakos Laboratories, Cellubril by AstraZeneca, and Laxicon by Stadmed.
Other uses
[edit]Dioctyl sodium sulfosuccinate is used as a surfactant in a wide range of applications, often under the name Aerosol-OT.[4][26] It is unusual in that it is able to form microemulsions without the use of co-surfactants, and it has a rich variety of aqueous-phase behavior including multiple liquid crystalline phases.[27]
Food additive
[edit]Dioctyl sodium sulfosuccinate has been approved by the US FDA as a "generally recognized as safe" (GRAS) additive.[28] It is used in a variety of food products, as a surface active agent, stabilizer, thickener, wetting agent, processing aid, solubilizing agent, emulsifier, and dispersant. The highest amount found in food products is 0.5% by weight, which include pasteurized cheese spreads, cream cheeses and salad dressings.[29] The FDA also approved its use as a wetting agent or solubilizer for flavoring agents in carbonated and non-carbonated drinks at levels up to 10 parts per million.[28]
Microencapsulation
[edit]Sodium docusate is the most widely used surfactant in reverse micelle encapsulation studies.[30]
Non-medical brand names
[edit]As a surfactant, docusate sodium is or has been commercialized under many brand names, including DSSj Aerosol OT, Alphasol OT, Colace, Complemix, Coprol, Dioctylal, Dioctyl-Medo Forte, Diotilan, Diovac, Disonate, Doxinate, Doxol, Dulsivac, Molatoc, Molofac, Nevax, Norval, Regutol, Softili, Solusol, Sulfimel DOS, Vatsol OT, Velmol, and Waxsol[31]
Chemistry
[edit]Structure and properties
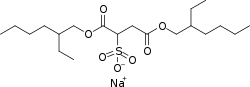
[edit]The structural formula of the docusate anion is R−O−C(=O)−CH(SO−3)−CH2−C(=O)−O−R, where R is the 2-ethylhexyl group H3C−(CH2)3−C(−CH2−CH3)H−CH2−. The conjugate acid can be described as the twofold carboxylate ester of sulfosuccinic acid with 2-ethylhexanol.
The compound is a white, wax-like, plastic solid, with an odor suggestive of octyl alcohol. It starts to decompose at about 220 °C.[31]
Solubility of dioctyl sodium sulfosuccinate in water is 14 g/L at 25 °C, increasing to 55 g/L at 70 °C.[31] Solubility is better in less polar solvents: 1:30 in ethanol, 1:1 in chloroform and diethylether, and practically unlimited in petroleum ether (25 °C). It also is highly soluble in glycerol, although this is a rather polar solvent. It is also highly soluble in xylene, oleic acid, acetone, diacetone alcohol, methanol, isopropanol, 2-butanol, methyl acetate, ethyl acetate, furfurol, and vegetable oils.[31]
The ester groups are easily cleaved under basic conditions, but are stable against acids.[18]
Synthesis
[edit]Sodium dioctyl sulfosuccinate can be obtained by treating dioctyl maleate with sodium bisulfite. The bisulfite anion adds to the double bond:
- −CH=CH− + HSO−3 → −CH(−SO−3)−CH2−
Toxicity
[edit]Ingestion may cause the side effects described above, such as diarrhea, intestinal bloating, and occasionally cramping pains. Dioctyl sodium sulfosuccinate is not known to be carcinogenic, mutagenic, or teratogenic.[32]
Marine species
[edit]Dioctyl sodium sulfosuccinate is of low toxicity for crustaceans such as the hermit crab Clibanarius erythropus and the shrimp Crangon crangon. Toxicity for molluscs varies widely, with 48-hour LD50 found between 5 mg/L for the common limpet and 100 mg/L for the common periwinkle. Various species of phytoplankton have an LD50 around 8 mg/L.
In a 2010 study, dioctyl sodium sulfosuccinate exhibited higher toxicity against bacteria (Vibrio fischeri, Anabaena sp.) and algae (Pseudokirchneriella subcapitata) than did a number of fluorinated surfactants (PFOS, PFOA, or PFBS). Measuring bioluminescence inhibition of the bacteria and growth inhibition of the algae, the LD50 were in the range of 43–75 mg/L. Combinations of the fluorinated compounds with dioctyl sodium sulfosuccinate showed mid to highly synergistic effects in most settings, meaning that such combinations are significantly more toxic than the individual substances.[33]
Freshwater species
[edit]The substance is highly toxic for rainbow trout with a median lethal concentration (LC50) of 0.56 mg/L after 48 hours for the pure substance. It is only slightly to moderately toxic for rainbow trout fingerlings, and slightly toxic for harlequin rasboras (LC50 27 mg/L of a 60% formulation after 48 hours).
References
[edit]- ^ a b c d e f g h "Docusate Salts". The American Society of Health-System Pharmacists. Archived from the original on 23 September 2015. Retrieved 11 August 2015.
- ^ a b American Society of Health-System Pharmacists (15 August 2011). "Stool Softeners". Archived from the original on 5 September 2015.
- ^ a b US 2181087, Caryl CR, Jaeger AO, "Detergent composition", issued 21 November 1939, assigned to American Cyanamid
- ^ a b c Wilson JL, Dickinson DG (May 1955). "Use of dioctyl sodium sulfosuccinate (aerosol O.T.) for severe constipation". Journal of the American Medical Association. 158 (4): 261–3. doi:10.1001/jama.1955.02960040019006a. PMID 14367076.
- ^ a b c Fakheri RJ, Volpicelli FM (February 2019). "Things We Do for No Reason: Prescribing Docusate for Constipation in Hospitalized Adults". Journal of Hospital Medicine. 14 (2): 110–113. doi:10.12788/jhm.3124. PMID 30785419.
- ^ a b c "Dioctyl Sulfosuccinate or Docusate (Calcium or Sodium) for the Prevention or Management of Constipation: A Review of the Clinical Effectiveness". CADTH Rapid Response Reports. 26 June 2014. PMID 25520993.
- ^ a b Candy B, Jones L, Larkin PJ, Vickerstaff V, Tookman A, Stone P (May 2015). "Laxatives for the management of constipation in people receiving palliative care" (PDF). The Cochrane Database of Systematic Reviews. 13 (5): CD003448. doi:10.1002/14651858.CD003448.pub4. PMC 6956627. PMID 25967924.
- ^ a b c d Ramkumar D, Rao SS (April 2005). "Efficacy and safety of traditional medical therapies for chronic constipation: systematic review". The American Journal of Gastroenterology. 100 (4): 936–71. doi:10.1111/j.1572-0241.2005.40925.x. PMID 15784043. S2CID 13869933.
- ^ a b c 2013 Nurse's Drug Handbook. Burlington, MA: Jones & Bartlett Learning. 2013. p. 366. ISBN 9781449642846.
- ^ Ash M, Ash I (2004). Handbook of preservatives. Endicott, N.Y.: Synapse information resources. p. 375. ISBN 9781890595661.
- ^ World Health Organization (2023). The selection and use of essential medicines 2023: web annex A: World Health Organization model list of essential medicines: 23rd list (2023). Geneva: World Health Organization. hdl:10665/371090. WHO/MHP/HPS/EML/2023.02.
- ^ a b "The Top 300 of 2022". ClinCalc. Archived from the original on 30 August 2024. Retrieved 30 August 2024.
- ^ "Docusate Drug Usage Statistics, United States, 2013 - 2022". ClinCalc. Retrieved 30 August 2024.
- ^ "Docusate Drug Usage Statistics, United States, 2013 - 2022". ClinCalc. Retrieved 30 August 2024.
- ^ Friedman M (October 1956). "Dioctyl sodium sulfosuccinate (doxinate) in chronic functional constipation". American Practitioner and Digest of Treatment. 7 (10): 1588–91. PMID 13362832.
- ^ "Docusate sodium". 18 December 2004. Archived from the original on 21 July 2011. Retrieved 6 March 2019.
- ^ Portalatin M, Winstead N (March 2012). "Medical management of constipation". Clinics in Colon and Rectal Surgery. 25 (1): 12–9. doi:10.1055/s-0032-1301754. PMC 3348737. PMID 23449608.
- ^ a b c d e f Dinnendahl V, Fricke U, eds. (2010). Arzneistoff-Profile (in German). Vol. 2 (23 ed.). Eschborn, Germany: Govi Pharmazeutischer Verlag. ISBN 978-3-7741-9846-3.
- ^ "How effective is docusate as a cerumenolytic agent?". GlobalRPH.com. Archived from the original on 23 November 2010.
- ^ Jasek W, ed. (2008). Austria-Codex Stoffliste (in German) (41st ed.). Vienna: Österreichischer Apothekerverlag. p. 316. ISBN 978-3-85200-190-6.
- ^ Yaffe SJ (2011). Drugs in pregnancy and lactation : a reference guide to fetal and neonatal risk (9th ed.). Philadelphia: Wolters Kluwer Health/Lippincott Williams & Wilkins. p. 1651. ISBN 9781608317080.
- ^ Mahadevan U, Kane S (July 2006). "American gastroenterological association institute medical position statement on the use of gastrointestinal medications in pregnancy". Gastroenterology. 131 (1): 278–82. doi:10.1053/j.gastro.2006.04.048. PMID 16831610.
- ^ drugs.com: Docusate Archived 16 July 2010 at the Wayback Machine
- ^ a b Shaw D (2017). "Drugs Acting on the Gastrointestinal Tract". Pharmacology and Therapeutics for Dentistry (Seventh ed.). pp. 404–416. doi:10.1016/B978-0-323-39307-2.00028-X. ISBN 9780323393072 – via Elsevier Science Direct.
- ^ Hamilton RJ (2013). Tarascon pocket pharmacopoeia : 2013 classic shirt-pocket edition (27 ed.). Burlington, Ma.: Jones & Bartlett Learning. p. 112. ISBN 9781449665869.
- ^ Whiffen AJ (1946). "Aerosol OT in the preparation of microscopic mounts of fungi". Mycologia. 38: 346. doi:10.1080/00275514.1946.12024063. PMID 20983186.
- ^ Nave S, Eastoe J, Penfold J (November 2000). "What Is So Special about Aerosol-OT? 1. Aqueous Systems". Langmuir. 16 (23): 8733–8740. doi:10.1021/la000341q.
- ^ a b "GRAS Notice Inventory Agency Response Letter GRAS Notice No. GRN 000006". Center for Food Safety and Applied Nutrition. 20 July 1998. Archived from the original on 31 October 2017. Retrieved 24 January 2020.
- ^ "CFR - Code of Federal Regulations Title 21". www.accessdata.fda.gov. Retrieved 29 January 2020.
- ^ Flynn PF (2004). "Multidimensional multinuclear solution NMR studies of encapsulated macromolecules". Prog. Nucl. Magn. Reson. Spectrosc. 45 (1–2): 31–51. doi:10.1016/j.pnmrs.2004.04.003.
- ^ a b c d Ahuja S, Cohen J (January 1973). "Dioctyl Sodium Sulfosuccinate". InAnalytical Profiles of Drug Substances. Vol. 2. Academic Press. pp. 199–219. doi:10.1016/S0099-5428(08)60040-4. ISBN 9780122608025.
- ^ ScienceLab.com: Docusate sodium Material Safety Data Sheet Archived 17 October 2006 at the Wayback Machine
- ^ Rosal R, Rodea-Palomares I, Boltes K, Fernández-Piñas F, Leganés F, Petre A (September 2010). "Ecotoxicological assessment of surfactants in the aquatic environment: combined toxicity of docusate sodium with chlorinated pollutants". Chemosphere. 81 (2): 288–93. Bibcode:2010Chmsp..81..288R. doi:10.1016/j.chemosphere.2010.05.050. PMID 20579683.
